 We are pleased that Dr. Michael Fisher has joined the FTCLDF website as a regular contributor. Dr. Fisher is a retired United States Department of Agriculture (USDA) Food Safety and Inspection Services (FSIS) veterinarian, bringing decades of experience enforcing FSIS regulations during the slaughter and processing of animals for which the USDA provides inspection services. Dr. Fisher is thrilled to bring you his expertise and guidance to help you navigate regulatory compliance. His goal is for small, USDA-inspected meat processors to succeed and to understand how to best maintain compliance and reduce regulatory issues.
We are pleased that Dr. Michael Fisher has joined the FTCLDF website as a regular contributor. Dr. Fisher is a retired United States Department of Agriculture (USDA) Food Safety and Inspection Services (FSIS) veterinarian, bringing decades of experience enforcing FSIS regulations during the slaughter and processing of animals for which the USDA provides inspection services. Dr. Fisher is thrilled to bring you his expertise and guidance to help you navigate regulatory compliance. His goal is for small, USDA-inspected meat processors to succeed and to understand how to best maintain compliance and reduce regulatory issues.
This month we consider a hazard analysis for the Thermally Processed—Commercially Sterile (TPCS) processing category. What is a TPCS product? If you do not know, you cannot conduct a hazard analysis.
- The Code of Federal Regulations (CFR) defines “product” to include any livestock or poultry carcass, part thereof, meat by-product, meat food product, or poultry food product capable of use as human food.[1]
- It is FSIS inspection policy that TPCS products are thermally processed, commercially sterile finished products in cans or flexible containers such as pouches, or semi-rigid, as in lunch bowls, prepared using commercial techniques described in 9 CFR 431.[2] TPCS products are shelf stable and can be safely stored at room temperature.[3] Classic examples of TPCS products are canned ham and chicken noodle soup.
What separates a TPCS product from Heat Treated – Shelf Stable product is the container. TPCS products are prepared in unopened, hermetically sealed containers using commercial techniques described in 9 CFR 431 that sterilize the product through the use of high temperatures that reduce or destroy microbial and enzyme activity and produce physical or chemical changes that make the product meet a certain quality standard. Heat Treated – Shelf Stable products incorporate curing, drying, or fermenting in combination with a heat treatment prior to placement in a sealed container.
TPCS products meet the standard of identify for ready-to-eat product.[4] What separates a TPCS product from other ready-to-eat products is that thermal processing destroys spore forming microbes not destroyed by 9 CFR 318.17 and 9 CFR 381.150 lethality treatments. Because thermal processing destroys spore forming microbes, a 9 CFR 318.17 and 9 CFR 381.150 stabilization is not required to achieve food safety.
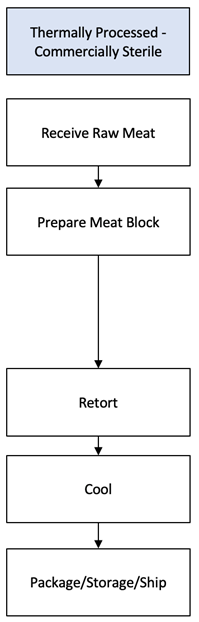
Now that we have identified a TPCS product, we need a 9 CFR 417.2(a)(2) flow chart describing the process steps and product flow for preparing the TPCS product. The diagram to the left is such a flow chart. Remember, a process is a series of steps conducted to an end, and a step is a unique activity that can encompass multiple actions. Our end is a TPCS product.
Grounds, facilities, equipment, utensils, chemicals, non-meat ingredients, and employees are potential sources of food safety hazards and common to all process steps. Maintaining 9 CFR 416 compliance prevents insanitary conditions and ensures that product is not adulterated by grounds, facilities, equipment, utensils, chemicals, non-meat ingredients, and employees. Therefore, a 9 CFR 417.1 preventive measure is not required to control a potential food safety hazard originating from grounds, facilities, equipment, utensils, chemicals, non-meat ingredients, and employees because it is already controlled by another regulatory performance standard. So, our hazard analysis makes no determination that a food safety hazard is reasonably likely to occur due to grounds, facilities, equipment, utensils, and employees during the TPCS process.
Receive Raw Meat: Raw product is a potential source of food safety hazard. A federally inspected, TPCS product must be prepared from federally inspected product. 9 CFR 318.1(a) and 9 CFR 381.145(a) require that such products be “prepared only in an official establishment and previously inspected and passed by a Program employee” to ensure they are not adulterated. The mark of inspection means that FSIS verified that the producing establishment followed the Hazard Analysis Critical Control Point (HACCP) process it determined is necessary to produce safe product.[5] USDA’s FSIS Office of Investigation Enforcement and Audit (OIEA) Compliance and Investigations Division conducts surveillance of product in commerce to assure that such product is not adulterated. An establishment that maintains 9 CFR 318.1(a), 9 CFR 381.145(a) and 9 CFR 416 compliance is sufficiently assured that product received is not adulterated. Therefore, our hazard analysis makes no determination that a food safety hazard is reasonably likely to occur during this process step.
Prepare Meat Block: Preparing the meat block can involve cutting, grinding, adding ingredients, mixing, injecting, or stuffing. I choose to capture these activities within a single process step. An official establishment may choose differently. TPCS products may contain ingredients, which are potential sources of food safety hazards. 9 CFR 318.6(a) requires that Ingredients not result in the product being adulterated. Non-meat and non-poultry ingredients are regulated by the Food and Drug Administration. FSIS accepts a guaranty by the ingredient supplier that the ingredient is safe for the intended use within the meaning of the Federal Food, Drug, and Cosmetic Act. A prudent establishment maintains such guaranty. No food safety hazard reasonably likely to occur carries over from the previous process step. Therefore, our hazard analysis makes no determination that a food safety hazard is reasonably likely to occur during this process step.
Retort: In the food industry, a retort is an autoclave; a machine used to carry out thermal processing of food on an industrial scale. Commercial retorts can process thousands of pounds of product in a single batch. The pressure cooker commonly used in households for canning fruits and vegetables is an autoclave.
The autoclave was invented in 1879. The technology was well advanced and recognized as producing safe foods when FSIS codified accepted industry practices in 1986.[6] Because the process was already codified when FSIS promulgated 9 CFR 417,[7] establishments preparing TPCS products need not address food safety hazard associated with microbiological contamination in their hazard analysis if the product is produced in accordance with 9 CFR 431.[8] It is in the establishment’s best interest to take advantage of this provision. There is one method to transform the meat block into a TPCS product: apply heat under pressure. Consultation with a process authority is necessary to determine the right combination of heat under pressure appropriate for the TPCS product prepared.
Cool: Unlike other ready-to-eat products, microbial growth is not a food safety concern during cooling. Because thermal processing destroys spore forming microbes, a 9 CFR 318.17 and 9 CFR 381.150 stabilization is not required to achieve food safety. Proper cooling of TPCS products are a product quality, not a food safety, concern.
Package/Storage/Ship: At this point in the process, the TPCS product is within an immediate container,[9] which may or may not bear a label, and may or may not be placed in a shipping container.[10] Because TPCS products are ready to eat, the 9 CFR 317.2(k) and 9 CFR 381.125(a) requirement to be labeled with Special Handling Statements (i.e., Keep Refrigerated, Keep Frozen, etc.), and the 9 CFR 317.2(k) and 9 CFR 381.125(a) requirements for Safe Handling Instructions do not apply.
Packaging, storage, and shipping are activities common to all processing categories, regardless of the product prepared. I choose to capture them within a single process step. An official establishment may choose differently. 9 CFR 317.24 or 9 CFR 381.144 and 9 CFR 325.1(c) or 9 CFR 381,190(c) require that packaging materials and transport methods, respectfully, not result in product being adulterated. An establishment that maintains 9 CFR 317.24 or 9 CFR 381.144 and 9 CFR 325.1(c) or 9 CFR 381,190(c) and 9 CFR 416 compliance is sufficiently assured that the product was not adulterated during packaging, storage, and shipment. No food safety hazard reasonably likely to occur carries over from the previous process step. Therefore, our hazard analysis makes no determination that a food safety hazard is reasonably likely to occur during this process step.
Hazard Analysis Complete! Key to conducting a TPCS hazard analysis is the realization that any hazard analysis rests on a foundation of sanitation and that there is a difference between what FSIS wants and what the regulations require. Absent that foundation of sanitation, conducing a TPCS hazard analysis is a waste of time because without sanitation, product that is not adulterated cannot be produced. Absent knowing the difference between what FSIS wants and what the regulations require, conducing a TPCS hazard analysis can be problematic because you are going against what the regulations require. Never a good idea.
Footnotes
[1] 9 CFR 301.2 and 9 CFR 381.1
[2] USDA, FSIS. (2019) FSIS Product Categorization (Guideline ID: FSIS-GD-2019-0010). https://www.fsis.usda.gov/guidelines/2019-0010
[3] USDA, FSIS. (2019) What does “shelf stable” mean? (Knowledge Article). https://ask.usda.gov
[4] 9 CFR 430.1: A ready-to-eat product is a meat or poultry product that is in a form that is edible without additional preparation to achieve food safety and may receive additional preparation for palatability or aesthetic, epicurean, gastronomic, or culinary purposes.
[5] Paul Kiecker, Acting FSIS Administrator (personal communication, April 12, 2018)
[6] USDA, FSIS, (1986). Canning of Meat and Poultry Products, Fed. Reg. Vol. 51, pgs. 45601-45647.
[7] USDA, FSIS, (1996). Pathogen Reduction; Hazard Analysis and Critical Control Point (HACCP) Systems, Fed. Reg. Vol. 61, pgs. 38805-38989.
[8] 9 CFR 417.2(b)(3): (3) HACCP plans for thermally processed/commercially sterile products do not have to address the food safety hazards associated with microbiological contamination if the product is produced in accordance with the requirements of part 431 of this chapter.
[9] 9 CFR 301.2: An immediate container is the receptacle or other covering in which any product is directly contained or wholly or partially enclosed. 9 CFR 381.1: An immediate container is any consumer package; or any other container in which poultry products, not consumer packaged, are packed.
[10] 9 CFR 301.2 and 9 CFR 381.1: A shipping container is the outside container (box, bag, barrel, crate, or other receptacle or covering) containing or wholly or partly enclosing any product packed in one or more immediate containers.
More
As always, if you have a question, please use the Contact Us link and ask.
Did you miss Dr. Fisher’s previous posts?
YOUR FUND AT WORK
Services provided by FTCLDF go beyond legal representation for members in court cases.
Educational and policy work also provide an avenue for FTCLDF to build grassroots activism to create the most favorable regulatory climate possible. In addition to advising on bill language, FTCLDF supports favorable legislation via action alerts and social media outreach.
You can protect access to real foods from small farms by becoming a member or donating today.